Introducción
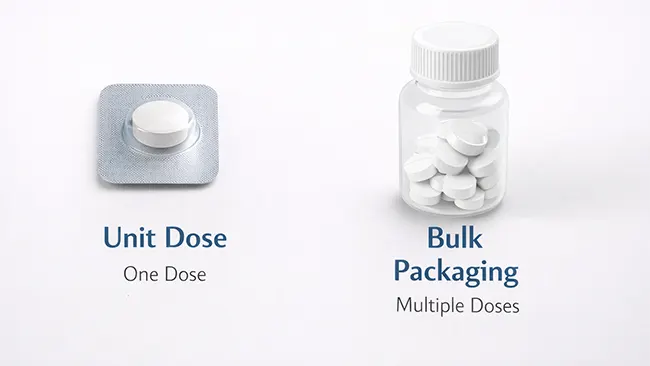
El envasado en dosis unitarias significa que una dosis viene envasada en una sola unidad lista para usar. En lugar de tomar el medicamento de un frasco grande o un envase a granel en el momento de su uso, la dosis ya está separada, envasada e identificada de antemano. En el envasado de medicamentos, esto generalmente significa que el medicamento se puede administrar directamente desde su envase como una sola unidad.
Un ejemplo sencillo es una tableta sellada en su propio blíster etiquetado. La misma idea también se puede aplicar a un pequeño paquete, vaso, vial u otra presentación de un solo uso preparada para una dosis. El formato del envase puede cambiar, pero el propósito básico sigue siendo el mismo: una dosis preparada, identificación clara y menos manipulación adicional antes de la administración.
Administración de Alimentos y Medicamentos de los Estados Unidos
)
Esto es importante porque el envasado en dosis unitarias no se limita a un tipo de máquina específico ni a un estilo de envase fijo. Se trata de un método de envasado que permite controlar cómo se presenta, etiqueta, manipula y protege cada dosis hasta su uso. La demanda de reenvasar productos orales sólidos en envases de dosis unitarias también ha aumentado en los entornos sanitarios, lo que explica por qué este término aparece con tanta frecuencia en las conversaciones sobre farmacia hospitalaria y envasado de medicamentos.
¿Qué es el envasado en dosis unitarias?
El envase monodosis está diseñado para una sola unidad de administración. El medicamento se envasa en un recipiente no reutilizable, que se utiliza directamente al administrar la dosis. Lo fundamental no es solo que el envase sea pequeño, sino que la dosis ya esté separada e identificada antes de su uso.
En la práctica, esto puede ser muy sencillo. Un comprimido en un blíster puede ser un envase de dosis unitaria. Una cantidad medida de líquido en un envase de un solo uso también puede ser un envase de dosis unitaria. Las directrices de las normas de farmacia hospitalaria añaden un detalle importante: un envase de dosis unitaria contiene una forma farmacéutica discreta, mientras que un envase de dosis unitaria contiene la dosis específica prescrita para el paciente. En muchos casos son lo mismo, pero no siempre. Un envase de dosis unitaria puede contener más de un comprimido si esa es la dosis que se debe administrar.
ASHP
)
Esto ayuda a aclarar un malentendido común. No todos los envases pequeños son verdaderos envases de dosis unitaria. Un envase compacto para venta al público puede contener la cantidad total de un tratamiento, en lugar de una sola unidad de administración. Los envases de dosis unitaria son más estrechos. Están diseñados para cada dosis individual y para contener la información necesaria para mantenerla claramente identificada y protegida hasta su uso.
Esa diferencia es importante para el envasado. Una vez que el producto se divide en dosis individuales, el envase debe permitir un etiquetado claro, una identificación legible y una protección fiable de la forma farmacéutica. Por lo tanto, cuando se habla de envases de dosis unitarias, no solo se hace referencia al tamaño del envase, sino también a la presentación y el control de la dosis.
Envases de dosis unitaria frente a envases de unidades de uso.
El envasado en dosis unitarias y el envasado en unidades de uso están relacionados, pero no son lo mismo. El envasado en dosis unitarias se organiza en torno a una sola administración a la vez. El envasado en unidades de uso se organiza en torno a la cantidad de dispensación completa en el envase original sellado del fabricante, generalmente suficiente para un tratamiento normal. La normativa sobre el etiquetado de dosis unitarias distingue claramente ambos conceptos, en lugar de considerarlos intercambiables.
La diferencia práctica es sencilla. El envase monodosis conserva la identidad de cada dosis individual durante su manipulación y administración. El envase de uso individual conserva la identidad del producto asociada al envase completo que se dispensa al paciente. Ambos pueden resultar prácticos y atractivos, pero su objetivo es diferente.
Esa diferencia también afecta al etiquetado. En un sistema de dosis unitaria, el envase individual debe contener suficiente información para que cada dosis separada sea reconocible y utilizable. En un envase de dosis unitaria, el envase completo lleva la identificación principal del tratamiento o la cantidad estándar que se dispensa. Por eso, ambos formatos pueden requerir diseños de etiquetas, lógicas de codificación y controles de manipulación diferentes, incluso cuando el producto en sí es el mismo.
Tipos de envases de dosis unitaria
El envasado en dosis unitarias no se limita a un solo formato. En la práctica, las formas comunes incluyen unidades basadas en blísteres para sólidos orales, paquetes pequeños o
paquetes de tiras
vasos utilizados en algunos procesos de reenvasado y envases líquidos de un solo uso, como viales o presentaciones similares. La idea central se mantiene incluso cuando cambia el envase inmediato: una dosis identificada se conserva como una sola unidad manipulada.
|
Formato
|
Uso típico
|
100%
|
|
Cavidad de ampolla
|
Comprimidos y cápsulas
|
Identidad del nivel de dosis, integridad del sellado, información celular legible
|
|
Tira o paquete
|
Sólidos orales reenvasados, algunos polvos
|
Sellado compacto, legibilidad del código, información clara del producto y del lote.
|
|
Vaso o presentación oral similar de un solo uso
|
Flujos de trabajo de farmacia o reenvasado
|
Control de manipulación, identificación legible, etiquetado de formato corto.
|
|
Envase de líquido de un solo uso
|
Líquidos orales y dosis líquidas medidas
|
Precisión del llenado, integridad del cierre, información legible del envase inmediato
|
Estos formatos no son iguales en todos los casos. Algunos funcionan mejor para una presentación clara de las tabletas, otros se eligen por su facilidad de reenvasado y otros por la necesidad de controlar una dosis líquida medida. La decisión sobre el formato no se basa tanto en la apariencia, sino en cómo se identificará, manipulará y administrará la dosis.

¿Por qué los hospitales utilizan envases de dosis unitaria?
Los hospitales utilizan envases monodosis porque la manipulación de medicamentos no se limita al almacenamiento, sino que también implica identificación, flujo de trabajo y administración. Al estar cada dosis separada y etiquetada, se reduce la manipulación adicional en el momento de la administración y la dosis se mantiene identificable hasta mucho más tiempo antes de su uso.
Evidencia reciente ayuda a explicar por qué este formato sigue siendo importante. Una revisión sistemática de 2025 sobre sistemas de dispensación de medicamentos en dosis unitarias en hospitales informó reducciones en los errores de medicación en todos los estudios incluidos, al tiempo que evaluó la rentabilidad. Un estudio de implementación hospitalaria de 2025 también encontró que las dosis unitarias piloto redujeron los errores de medicación y de procedimiento y mejoraron el escaneo de códigos de barras dentro del proceso de medicación. Estos hallazgos no significan que todos los programas de dosis unitarias funcionen de la misma manera, pero sí respaldan el valor práctico del envasado a nivel de dosis en entornos hospitalarios.
PubMed
)
El uso de códigos de barras forma parte de este panorama. Las directrices federales sobre códigos de barras explican que estos ayudan a los profesionales sanitarios a verificar que se administra el medicamento correcto, en la dosis y vía de administración adecuadas, al paciente correcto y en el momento preciso. Cuando los envases monodosis incluyen una identificación legible y escaneable a nivel de dosis individual, se adaptan mejor al flujo de trabajo que los envases más grandes, que aún requieren una posterior separación o interpretación.
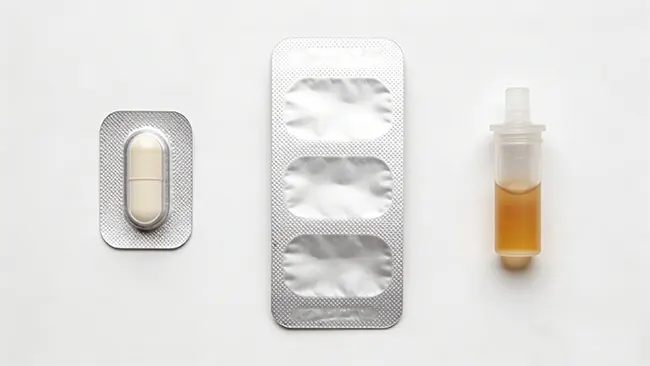
Envases en blíster monodosis, sobres y formatos líquidos.
Envase tipo blíster
Es uno de los ejemplos más claros de envases monodosis para sólidos orales. Separa físicamente cada comprimido o cápsula, protege la dosis hasta su uso y proporciona al envase un espacio definido para la información de identificación y control. Las directrices de seguridad para envases tipo blíster recomiendan que la información clave, como el nombre del producto, la concentración, el número de lote, la fecha de caducidad, el código de barras y el fabricante, permanezca visible sobre cada compartimento del blíster siempre que sea posible, especialmente si los compartimentos pueden separarse del envase exterior.
Los paquetes individuales y los envases tipo tira cumplen una función similar en algunos procesos de reenvasado. La estructura del material y el patrón de sellado difieren de los de un blíster, pero la lógica del envase es la misma. Cada paquete individual debe contener una dosis de forma clara para que pueda manipularse como una unidad individual y no como parte de un suministro mayor e indiviso.
Los formatos líquidos añaden un nivel adicional de control. Una dosis líquida medida en un envase monodosis puede funcionar bien por la misma razón que un blíster para una tableta: la dosis se prepara con antelación, se limita a una sola administración y depende menos de mediciones o vertidos posteriores. Sin embargo, el desafío del envasado es diferente, ya que las dosis unitarias líquidas dependen en mayor medida de la precisión del llenado, la integridad del cierre y la legibilidad de la información en un envase inmediato mucho más pequeño.
Equipos de envasado utilizados para formatos de dosis unitarias
El equipo utilizado para el envasado en dosis unitarias depende del formato del envase inmediato. Para sólidos orales envasados en una dosis por cavidad, un
Máquina de envasado en blíster
La máquina de envasado en blíster (o similar) suele ser la más adecuada, ya que permite la presentación a nivel de cavidad, el sellado y la identificación dosis por dosis. En este formato, la tarea de envasado no se limita a formar y sellar el paquete, sino que también debe garantizar que cada unidad sea legible y reconocible tras su manipulación, separación o distribución.
Para presentaciones en tiras o paquetes, se necesita una
Máquina de embalaje en tiras
Por lo general, es una mejor opción. Estos formatos dependen de un sellado compacto y una codificación clara en una superficie muy limitada, por lo que la ubicación de la impresión y la legibilidad del código son tan importantes como la forma básica del envase. Cuando cada envase contiene una dosis, la información del lote, los detalles de la fecha de caducidad y la identidad del producto deben permanecer claros incluso en un envase pequeño de uso inmediato.
Para dosis unitarias líquidas, las prioridades de los equipos cambian de nuevo.
máquina de llenado de líquidos
Los envases pequeños de un solo uso deben garantizar un control preciso del llenado, un cierre seguro y una identificación legible en una pequeña área de la etiqueta. En comparación con los formatos de dosis sólidas, las presentaciones líquidas exigen mayor atención a la prevención de fugas y a la manipulación del envase, ya que este debe proteger una dosis medida sin generar problemas de legibilidad o integridad posteriormente.
Después
embalaje primario
, el equipo de etapa posterior puede incluir una máquina etiquetadora, un sistema de inspección visual y
máquina de encartonado
, dependiendo de cómo se verifique y empaquete el producto para su distribución. Una vez que el producto se ha separado en unidades de dosis, cada paso posterior debe preservar la legibilidad, la trazabilidad y la integridad del empaque.

Máquina de blíster y máquina de embalaje en tiras
Envases monodosis frente a envases multidosis
La diferencia más clara entre los envases monodosis y los multidosis radica en el nivel de organización del envase. Los monodosis se organizan en torno a una sola unidad de administración. Los envases multidosis se organizan en torno a un suministro mayor que se abrirá, contará, verterá o utilizará repetidamente a lo largo del tiempo. Esta diferencia fundamental modifica la manipulación, la presión del etiquetado y el diseño del flujo de trabajo.
En un envase multidosis, el recipiente lleva la identificación del producto para la mayor cantidad. En un envase monodosis, el envase individual debe contener la información que permite identificar cada dosis por separado. Esto suele aumentar las exigencias de envasado y etiquetado del envase individual, pero puede reducir la manipulación adicional durante el proceso de uso del medicamento.
|
Punto
|
Envase monodosis
|
Envase multidosis
|
|
Enfoque del paquete
|
Una unidad administrativa
|
Mayor suministro
|
|
Identidad de empaque inmediato
|
Alta importancia
|
Importancia del nivel de dosis más bajo
|
|
Manipulación adicional antes de su uso
|
Más bajo
|
Generalmente más alto
|
|
Demanda de envases por dosis
|
Más alto
|
Más bajo
|
|
Mejor ajuste
|
Flujos de trabajo hospitalarios y de dosis controladas
|
Suministro estándar y acceso repetido
|
Esto no significa que un formato sea universalmente mejor que el otro. La mejor opción depende del producto, el entorno asistencial, el nivel de detalle del etiquetado requerido y el grado de control de la dosis que necesita el flujo de trabajo. La experiencia hospitalaria respalda el valor de los sistemas de dosis unitarias, pero también demuestra que la calidad de la implementación y el diseño del proceso siguen siendo importantes.
Conclusión
El envasado en dosis unitarias se entiende mejor como un enfoque de envasado por niveles de dosis, no como un único tipo de envase fijo. Un blíster, un envase en tira, un paquete o un recipiente líquido monodosis pueden ajustarse a este concepto si el envase está diseñado para una unidad de administración y mantiene la dosis identificable hasta su uso.
Por eso, este tema se sitúa en la intersección del envasado, el etiquetado y la manipulación de medicamentos. El formato elegido para el envasado en dosis unitarias modifica la función del envase inmediato, la forma de identificar la dosis y los equipos de envasado y los pasos de inspección más importantes. Una vez comprendida esta lógica, resulta mucho más fácil entender la diferencia entre el envasado en dosis unitarias, en unidades de uso y en dosis múltiples.
Preguntas frecuentes
¿Qué es el envasado en dosis unitarias?
El envasado en dosis unitarias significa que una dosis se envasa como una unidad de administración lista para usar en un recipiente inmediato no reutilizable, de modo que el medicamento se puede administrar directamente desde ese envase.
¿El envase tipo blíster siempre es un envase monodosis?
No siempre. Un blíster puede funcionar como envase de dosis unitaria cuando cada cavidad representa una unidad de administración y contiene la información de identificación y control necesaria, pero no todas las presentaciones en blíster deben tratarse automáticamente como un formato de dosis unitaria real.
¿Por qué los hospitales prefieren los envases monodosis?
Los hospitales suelen utilizarlo porque facilita la identificación de la dosis, reduce la manipulación innecesaria y se integra bien con los flujos de trabajo de medicación asistidos por códigos de barras. Estudios recientes también han reportado reducciones en los errores de medicación y de procedimiento tras la implementación de la dosis unitaria.
¿Qué información debe aparecer en un envase de dosis unitaria?
Los requisitos exactos varían según el producto y el contexto, pero la identidad, la potencia, la información de lote o control, la fecha de caducidad y la legibilidad del código de barras son aspectos fundamentales. En el caso de los blísteres, las directrices de seguridad también destacan la importancia de mantener la información clave disponible a nivel de celda siempre que sea posible.
¿El envasado en dosis unitarias requiere un equipo diferente al del envasado en dosis múltiples?
A menudo sí. La línea exacta depende del formato, pero el envasado a nivel de dosis generalmente exige mayor legibilidad inmediata del envase, colocación del código, inspección e integridad del envase, ya que cada unidad separada debe permanecer identificable y utilizable por sí sola.
Referencias
●
Guía de la FDA sobre el etiquetado de dosis unitarias para formas farmacéuticas orales sólidas y líquidas.
Administración de Alimentos y Medicamentos de los Estados Unidos
)
●
Guía de ASHP sobre envases de dosis única y monodosis de medicamentos.
ASHP
)
●
Evidencia reciente de hospitales sobre la dispensación de dosis unitarias, errores de medicación y flujo de trabajo con códigos de barras.
PubMed
)

































 Red IPv6 admitida
Red IPv6 admitida