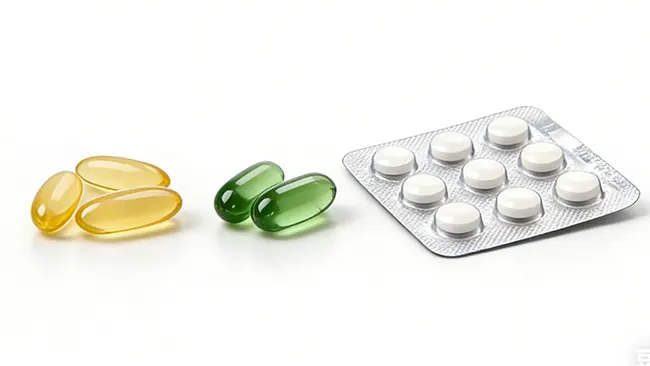
Una forma farmacéutica sólida es una de las maneras más comunes de fabricar y administrar medicamentos y suplementos. Las tabletas, cápsulas, polvos y gránulos entran en esta categoría. Para fabricantes, compradores y planificadores de proyectos novatos, esto es importante porque la forma farmacéutica no solo afecta la apariencia del producto o la forma en que el usuario lo toma, sino también las opciones de formulación, las etapas de producción, las vías de envasado, las condiciones de almacenamiento y la selección de equipos. La guía de la OMS describe las formas farmacéuticas sólidas orales como un grupo amplio que abarca desde polvos hasta tabletas recubiertas, y destaca ventajas típicas como una mayor estabilidad, una buena uniformidad de la dosificación y un envasado y transporte convenientes.
Por eso, la forma farmacéutica sólida es un concepto de partida útil. Mucha gente se fija primero en el producto terminado, como un comprimido en un blíster o una cápsula en un frasco. Pero detrás de ese producto terminado hay toda una lógica de fabricación. Un polvo que requiere un llenado preciso se comporta de forma diferente a un comprimido que debe comprimirse y, a veces, recubrirse. Una línea de encapsulado tiene requisitos de proceso distintos a los de una línea de llenado de gránulos. Cuando cambia la forma farmacéutica, el proceso de producción suele cambiar también.
Para quienes se inician en este tema, puede resultar confuso debido al uso impreciso de ciertos términos. Algunos utilizan el término «tableta» como si se refiriera a todos los medicamentos orales, mientras que otros emplean «cápsula» como término genérico para cualquier sustancia que se ingiera por vía oral. En la práctica, las formas farmacéuticas sólidas abarcan una gama más amplia de formatos, cada uno con sus propias ventajas, limitaciones e implicaciones en el envasado.
Para un fabricante farmacéutico o nutracéutico, elegir el formato adecuado rara vez es solo una decisión de marketing. Generalmente implica equilibrar la precisión de la dosis, el flujo del material, la estabilidad, la sensibilidad a la humedad, la facilidad de deglución, los objetivos de producción y el estilo de envasado. Comprender primero esta categoría facilita enormemente las decisiones posteriores sobre la selección de maquinaria, el diseño de la línea y el envasado.

¿Qué es una forma farmacéutica sólida?
Una forma farmacéutica sólida es un producto farmacéutico o nutracéutico que se presenta en estado sólido, con el principio activo y los excipientes preparados en cantidades medidas para su uso. En términos sencillos, significa que el producto no se presenta en forma líquida, suspensión, crema o semisólida. En cambio, se elabora y suministra como una unidad sólida o en una cantidad medida en estado sólido.
Los ejemplos más conocidos son las tabletas y las cápsulas, pero el concepto es más amplio. Los polvos y los gránulos también son formatos comunes en esta categoría, especialmente cuando el producto se va a envasar en sobres, stick packs, botellas o envases monodosis. En algunos casos, los pellets, las pastillas y formas orales similares también pueden pertenecer a este grupo más amplio, dependiendo del diseño y la aplicación del producto. La terminología de formas farmacéuticas de la FDA reconoce por separado formas como cápsulas y tabletas, mientras que la guía de la OMS incluye explícitamente polvos, tabletas recubiertas, tabletas masticables, tabletas de disolución oral, gránulos, pellets y minitabletas dentro de las discusiones sobre formas farmacéuticas sólidas orales.
Administración de Alimentos y Medicamentos de los Estados Unidos
)
Cuando se habla de dosis sólidas orales, generalmente se hace referencia a productos sólidos que se ingieren por vía oral. Esto incluye comprimidos, cápsulas duras, cápsulas blandas, gránulos y polvos. Si bien el término general abarca toda la categoría, la dosis sólida oral se usa a menudo cuando se hace referencia específicamente a productos que se tragan, mastican, disuelven en la boca o se mezclan antes de tomarlos.
Una de las razones por las que esta categoría es tan importante es que los formatos sólidos se utilizan ampliamente tanto en productos farmacéuticos como en suplementos. Suelen ser más fáciles de dosificar de forma consistente, transportar y envasar a gran escala que muchos formatos líquidos. Además, se integran bien con los procesos industriales establecidos, razón por la cual muchas líneas de producción se basan en ellos. La OMS destaca específicamente la mayor estabilidad, la buena uniformidad de la dosificación y la facilidad de transporte y envasado como ventajas clave de las formas farmacéuticas orales sólidas.
Sin embargo, no todos los formatos de esta categoría se comportan igual en la fabricación. Un comprimido suele depender de la compresión. Una cápsula depende del manejo de la cubierta y la consistencia del relleno. Un producto en polvo o granulado puede no necesitar compresión, pero sí requerir mayor atención a la precisión de la dosificación, la fluidez y el control de la humedad. Por eso, comprender la definición es solo el primer paso. Una vez elegido el formato, el proceso de producción se vuelve mucho más claro.
Tipos comunes de formas farmacéuticas sólidas
La mayoría de los compradores piensan primero en tabletas y cápsulas, pero la categoría es más amplia que eso.
tabletas
Los comprimidos son unidades sólidas comprimidas diseñadas para administrar una dosis definida en un formato compacto. Pueden ser simples, recubiertos, masticables, dispersables o de liberación modificada, según el diseño del producto. Los comprimidos son populares porque generalmente permiten una alta producción, un control preciso de la dosis y un envasado posterior eficiente.
Cápsulas
Las cápsulas son otro formato común en esta categoría, especialmente para polvos, gránulos y algunos rellenos líquidos o semisólidos dentro de una cubierta. Las cápsulas duras son comunes tanto en productos farmacéuticos como en suplementos. Suelen elegirse cuando la fórmula no se presta bien a la compresión o cuando el equipo de producto prefiere una presentación en cápsula por razones comerciales.
Polvos y gránulos
En este tema, los polvos y gránulos suelen subestimarse, pero constituyen formatos importantes dentro de esta categoría. Pueden presentarse en sobres, envases individuales, frascos o cápsulas. En muchos productos, los gránulos mejoran la fluidez, la manipulación y la dosificación en comparación con el polvo suelto. Las directrices de la OMS abordan específicamente los polvos y las partículas múltiples, como los gránulos y los pellets, incluyendo su uso en sobres o cápsulas duras.
Otras formas sólidas comunes
Según el producto, otras presentaciones pueden incluir gránulos, minicomprimidos, pastillas o comprimidos bucodispersables. No es necesario que todos los sitios web las abarquen en detalle, pero ayudan a demostrar que se trata de una categoría amplia, no de una única presentación.
|
Forma farmacéutica
|
Presentación típica
|
Puntos fuertes principales
|
Ruta de fabricación común
|
Ruta de embalaje típica
|
|
Tableta
|
Unidad de compresión
|
Alto rendimiento, dosis precisa, fácil conteo.
|
Mezclado → granulación o compresión directa → prensa de comprimidos → recubrimiento opcional
|
Envases blíster, botellas, cajas
|
|
Cápsula
|
Vaina rellena de polvo, pastillas o gránulos.
|
Tipo de llenado flexible, formato familiar para el consumidor.
|
Mezclado o granulación → llenado de cápsulas → pulido/inspección
|
Envases blíster, botellas, cajas
|
|
Polvo
|
Producto de medida suelta
|
Peso de relleno flexible, sin paso de compresión
|
Mezcla → dosificación/llenado
|
Sobres, paquetes individuales, botellas
|
|
Gránulo
|
Partículas de flujo libre
|
Mejor manejo que el polvo suelto, flexibilidad de dosificación.
|
Mezclado → granulación → secado/clasificación → llenado o procesamiento posterior
|
Sobres, paquetes individuales, botellas, cápsulas
|

¿Por qué se utilizan tanto las formas farmacéuticas sólidas?
Existe una razón sencilla por la que las formas farmacéuticas sólidas siguen siendo las predominantes: satisfacen las necesidades de fabricación y del mercado al mismo tiempo.
Desde el punto de vista del producto, suelen ofrecer una buena consistencia en la dosis y una vida útil práctica. Desde el punto de vista logístico, generalmente son más fáciles de almacenar, contar, enviar y exhibir que muchos formatos líquidos. Desde el punto de vista de la fabricación, se adaptan bien a los procesos establecidos a gran escala. La OMS destaca la estabilidad, la uniformidad de la dosis, la facilidad de envasado y la facilidad de transporte como ventajas clave de las formas farmacéuticas orales sólidas.
También ofrecen flexibilidad. Una marca puede optar por tabletas para optimizar la producción y los costos, cápsulas para mayor flexibilidad de llenado, o polvos y gránulos para envases monodosis o mezclas para bebidas. Esta flexibilidad es una de las razones por las que esta categoría sigue siendo útil en lugar de convertirse en una etiqueta técnica restrictiva.
Cómo afectan las formas farmacéuticas sólidas a la fabricación
Una vez elegida la forma farmacéutica, el proceso de fabricación suele volverse más estructurado.
La fabricación de comprimidos suele implicar la preparación del polvo, el control del flujo, la compresión y, en ocasiones, el recubrimiento. La fabricación de cápsulas generalmente se centra en la calidad de la mezcla, la consistencia del peso de llenado, la separación de las cápsulas, el cierre de la tapa y el embalaje. La fabricación de polvos o gránulos puede eliminar la compresión del proceso, pero aumenta la atención a la precisión de la dosificación, la fluidez, el control del polvo y la compatibilidad del sellado en el envase final.
Aquí es donde los principiantes suelen subestimar el impacto de la forma farmacéutica. No se trata solo de una elección del producto final, sino que también influye en el tipo de control de procesos necesario en la línea de producción. La guía ICH Q8 explica que los atributos críticos de calidad para las formas farmacéuticas orales sólidas suelen estar relacionados con la pureza, la potencia, la liberación del fármaco y la estabilidad, y que las propiedades del material, como la distribución del tamaño de partícula y la densidad aparente, pueden afectar la calidad del producto final. La guía de inspección de la FDA para formas farmacéuticas orales sólidas también enfatiza que el desarrollo del proceso, las especificaciones y la validación deben establecerse antes de la producción rutinaria a gran escala.
Para un comprador de equipos, esto importa porque los diferentes formatos de producto requieren diferentes máquinas centrales. Los proyectos de tabletas pueden centrarse en una
prensa rotativa para tabletas
, desempolvador, detector de metales y equipo de recubrimiento. Los proyectos de cápsulas pueden centrarse en una máquina de llenado de cápsulas, pulidora de cápsulas y pasos de clasificación o inspección. Los proyectos de polvo y gránulos pueden depender más de mezcladores, granuladores, secadores, sistemas de llenado de tornillo sin fin,
máquinas de paquetes de varillas de múltiples carriles
o máquinas de envasado en sobres.
Cómo la forma farmacéutica influye en el embalaje y los equipos.
El empaquetado no es una decisión que se toma al final. En muchos casos, la forma farmacéutica ya indica la posible vía de envasado.
Las tabletas y cápsulas a menudo se dirigen hacia
envase blíster
o líneas de conteo y embotellado. Los polvos y gránulos suelen envasarse en sobres, paquetes individuales, bolsas o frascos, según el tamaño de la dosis y la presentación comercial. La OMS señala que los polvos y las preparaciones multiparticuladas pueden presentarse en sobres o cápsulas duras, lo que nos recuerda que una misma fórmula puede tener diferentes destinos en la cadena de suministro.
Por eso, un comprador no debería elegir primero la forma farmacéutica y preocuparse después por el envase. Un comprimido sensible a la humedad puede requerir un blíster con mayor barrera. Un producto en cápsulas destinado a la venta al por menor puede requerir una configuración de procesamiento diferente a la de la misma cápsula vendida en blísteres. Un producto granulado de dosis única puede ser mucho más adecuado para un envase tipo stick pack que para un frasco.
Para un fabricante, aquí es donde la planificación de equipos se vuelve más práctica. La pregunta no es solo "¿Qué producto queremos?", sino también "¿Qué línea de producción soportará esta forma farmacéutica de manera eficiente, desde la alimentación hasta el envasado final?".
Errores comunes al evaluar una forma farmacéutica sólida
Un error común entre los principiantes es considerar todos estos formatos como intercambiables. No lo son. Dos productos pueden parecer igual de sencillos en un estante, pero comportarse de forma muy diferente durante la producción.
Otro error común es centrarse únicamente en el formato para el consumidor. Una cápsula puede parecer atractiva en el mercado, pero la fórmula podría ser más eficaz en forma de tableta. Un polvo puede parecer sencillo, pero su mala fluidez y la absorción de humedad pueden generar problemas de llenado y envasado si la vía de administración no se elige correctamente.
Un tercer error es elegir las máquinas una por una en lugar de pensar en lógica lineal. Una prensa de tabletas,
llenador automático de cápsulas
La máquina de blíster o la línea de embotellado no deben seleccionarse de forma aislada. La forma farmacéutica conecta la preparación previa, la dosificación o el formado principal, la inspección, el envasado y el empaquetado final.
Cómo elegir entre las formas farmacéuticas sólidas más comunes
La elección óptima depende de las prioridades fundamentales de su producto.
Si la prioridad es una alta producción y una fabricación eficiente en grandes lotes, los comprimidos suelen ser una opción atractiva. Si la fórmula es difícil de comprimir o el mercado prefiere la presentación en cápsulas, estas pueden ser más convenientes. Si el producto se concibe como una bebida preparada, un polvo oral monodosis o una presentación con peso de llenado flexible, los polvos y gránulos pueden ser más adecuados.
La estabilidad también es importante. Algunos productos son más sensibles a la humedad, el oxígeno o la presión durante su manipulación. Otros requieren una presentación que facilite su ingestión, enmascare el sabor o permita su consumo en dosis individuales. En estos casos, la mejor opción es aquella que equilibra el rendimiento del producto con un proceso de fabricación y envasado viable.
Para los compradores que aún están en las primeras etapas del proceso, una forma práctica de comparar opciones es hacerse cuatro preguntas:
1. ¿Qué forma se ajusta mejor a la fórmula?
2. ¿Qué formato se adapta mejor al mercado objetivo?
3. ¿Qué forma se puede fabricar de manera consistente a la escala prevista?
4. ¿Qué forma se ajusta a la ruta de embalaje prevista con la menor fricción?
Este enfoque suele dar como resultado una decisión mucho mejor que partir únicamente de la apariencia del producto.
Conclusión
Una forma farmacéutica sólida no es un único tipo de producto. Es una categoría amplia que incluye comprimidos, cápsulas, polvos, gránulos y otras presentaciones sólidas utilizadas en la fabricación de productos farmacéuticos y nutracéuticos.
Por eso este tema es importante. Elegir el formato adecuado no solo afecta la apariencia final del producto, sino que también influye en la formulación, los pasos de producción, el empaquetado y la planificación del equipo. Para un comprador principiante, comprender esta relación desde el principio facilita enormemente las decisiones posteriores.
Preguntas frecuentes
¿Qué es una forma farmacéutica sólida?
Una forma farmacéutica sólida se refiere a un producto farmacéutico o nutracéutico que se presenta en estado físico sólido, incluyendo tabletas, cápsulas, polvos y gránulos.
¿Cuáles son algunos ejemplos comunes de formas farmacéuticas sólidas?
Algunos ejemplos comunes son las tabletas, las cápsulas, los polvos, los gránulos, los comprimidos y algunas formas dispersables por vía oral.
¿Son las cápsulas una forma farmacéutica sólida?
Sí. Las cápsulas son ampliamente reconocidas como una forma farmacéutica sólida común, especialmente para polvos, gránulos y pastillas.
¿Cuál es la diferencia entre una forma farmacéutica sólida y una forma farmacéutica sólida oral?
La forma farmacéutica sólida es la categoría más amplia. La forma farmacéutica sólida oral generalmente se refiere específicamente a las formas sólidas que se toman por vía oral.
¿Qué forma farmacéutica sólida es la más fácil de fabricar?
No existe una única respuesta. Los comprimidos pueden ofrecer resultados muy elevados, pero las cápsulas pueden ser más adecuadas para algunas formulaciones. Los polvos y gránulos pueden simplificar un paso y complicar otro. La opción más sencilla depende del comportamiento de la fórmula, la dosis objetivo y el plan de envasado.
¿Cómo influye la forma farmacéutica en la elección del envase?
La forma farmacéutica suele determinar el tipo de envase. Los comprimidos y las cápsulas se suelen envasar en blísteres o frascos, mientras que los polvos y los gránulos se suelen envasar en sobres, envases individuales o frascos.
Referencias
●
Organización Mundial de la Salud. Desarrollo de medicamentos pediátricos: aspectos a considerar en el desarrollo farmacéutico — sección sobre formas farmacéuticas orales sólidas. (
Organización Mundial de la Salud
)
●
ICH Q8(R2). Desarrollo farmacéutico: análisis de los atributos críticos de calidad para las formas farmacéuticas orales sólidas. (
ICH
)






























 Red IPv6 admitida
Red IPv6 admitida